
“The remedy has actually reworked my life greater than I might have ever imagined,” Victoria Grey, the primary particular person to obtain the CRISPR gene-editing therapy tellls NPR. “It gave me a brand new lease on life.”
Orlando Gili for NPR
disguise caption
toggle caption
Orlando Gili for NPR

“The remedy has actually reworked my life greater than I might have ever imagined,” Victoria Grey, the primary particular person to obtain the CRISPR gene-editing therapy tellls NPR. “It gave me a brand new lease on life.”
Orlando Gili for NPR
4 years in the past, Victoria Grey was mendacity in a hospital mattress, exhausted by a lifetime with sickle cell illness and recovering from a grueling experimental therapy she hoped would save her.
Immediately, the Forest, Miss., mom of 4 is brimming with power and hope. She’s working full time, maintaining along with her teenage kids and touring the world along with her husband.
Grey’s transformation is the results of the landmark medical experiment that she launched and which culminated in December with a milestone: The Meals and Drug Administration’s accredited the primary therapy that makes use of gene-editing to alleviate a human sickness. It is the primary genetic remedy for the brutal blood dysfunction, which had lengthy been uncared for by drugs.
For the previous 4 years, NPR has been chronicling Grey’s story. NPR broke the information when Grey turned the primary affected person to get the therapy and had unique entry to report on her ensuing expertise.
“I am ecstatic,” Grey informed NPR in regards to the Dec. 8 approval. “It is a blessing that they accredited this remedy. It is a new starting for folks with sickle cell illness.”
A medical marvel that could be out of attain
Whereas the therapy has turned Grey and different sufferers’ lives round, many hurdles stay. The remedy is dear — $2.2 million per affected person — and is difficult and arduous, elevating questions on whether or not it is going to be accessible to the sufferers who want it most. Sickle cell disproportionately impacts folks of African descent and is most typical in much less prosperous international locations.
Different questions stay as nicely: Will the therapy hold working? Will it prolong sufferers’ lifespans? Will it trigger unexpected issues sooner or later?
“That is one thing that we have been ready for greater than 70 years,” stated Dr. Lewis Hsu on the College of Illinois Chicago, who serves because the chief medical officer of the Sickle Cell Affiliation of American. “However there are various points forward of us.”
On the similar time, the approval is producing pleasure as a result of different remedies that use the gene-editing method CRISPR are displaying promise for different ailments, starting from uncommon however devastating genetic issues, akin to muscular dystrophy, to frequent illnesses, together with most cancers and coronary heart illness.
“It is solely the start,” stated Jennifer Doudna of the College of California, Berkeley, who shared a Nobel Prize for serving to uncover CRISPR. CRISPR permits scientists to make very exact modifications in DNA far more simply than ever earlier than. “It is an incredible time,” she informed NPR.
A grueling therapy for a brutal illness
When NPR launched Grey to listeners and readers in July 2019, she was 34 and had simply undergone the gene-editing process on the Sarah Cannon Analysis Institute in Nashville, Tenn. It was uncharted territory, spurring excessive hopes but in addition fraught with uncertainty and threat. It was the primary time cells that had been edited with CRISPR had been infused right into a affected person with sickle cell.
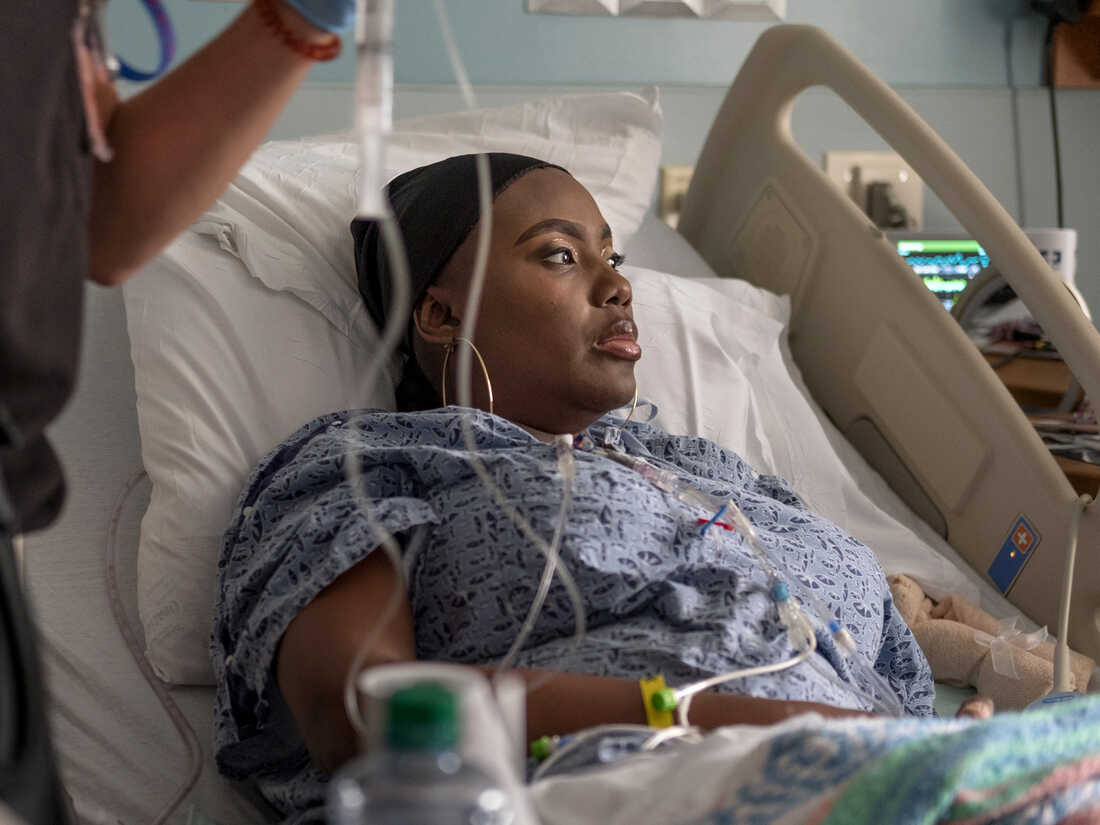
Victoria Grey volunteered for some of the anticipated medical experiments in a long time: the primary try to make use of the gene-editing method CRISPR to deal with a genetic dysfunction within the U.S.
Meredith Rizzo/NPR/Meredith Rizzo/NPR
disguise caption
toggle caption
Meredith Rizzo/NPR/Meredith Rizzo/NPR

Victoria Grey volunteered for some of the anticipated medical experiments in a long time: the primary try to make use of the gene-editing method CRISPR to deal with a genetic dysfunction within the U.S.
Meredith Rizzo/NPR/Meredith Rizzo/NPR
Sickle cell impacts hundreds of thousands of individuals world wide, together with about 100,000 within the U.S. A genetic defect causes bone marrow to supply a faulty model of the protein referred to as hemoglobin. That creates deformed, sickle-shaped pink blood cells that jam blood vessels and do not carry oxygen usually, inflicting debilitating and finally life-shortening issues.
“It is horrible. When you possibly can’t stroll or, you realize, elevate up spoon to feed your self, it will get actual onerous,” Grey stated on the time.
One of many worst results of sickle cell are unpredictable assaults of excruciating ache. These crises drive sufferers to hurry to the hospital for emergency blood transfusions and highly effective narcotics to alleviate the ache.
For the gene-editing therapy, medical doctors take away cells from sufferers’ bone marrow so scientists can use CRISPR to edit a gene, enabling the cells to make a wholesome type of hemoglobin referred to as fetal hemoglobin. Sufferers then bear chemotherapy to make room of their bone marrow for the edited cells. Lastly, medical doctors infuse billions of the modified cells again into sufferers’ our bodies.
NPR produced a collection of unique stories that documented Grey’s progress. That included when she had recovered sufficient from the July 2, 2019, process to depart Nashville months later to make a shock return house to her household in Mississippi.

“I do know it will be emotional for me. I miss the hugs and the kisses and simply every little thing,” she stated then.
NPR was additionally there when Grey traveled again to Nashville frequently over the next months so medical doctors might see if the therapy was working.
Early indicators of success
Throughout her November 2019 analysis in Nashville, Grey’s physician revealed the thrilling information that he had discovered the primary indicators that the therapy could also be profitable — her genetically modified “supercells” — as Grey referred to as them — had been producing fetal hemoglobin.
“It is a miracle,” Grey stated on the time. “While you pray for one thing for thus lengthy, all you possibly can have is hope. It is wonderful.”
Getting that information simply earlier than holidays made it notably particular “as a result of generally I might be within the hospital on Christmas” as an alternative of along with her household, she stated. “I am wanting ahead to an entire new life for all of us.”

Over the subsequent 4 years, it turned clear that the therapy was, actually, working. Grey by no means skilled one other ache crises or the debilitating fatigue that had plagued her all her life.
“It is great. It is the change I have been ready on my complete life,” Grey informed NPR a yr after getting handled.
The change enabled Grey to take care of herself and her kids for the primary time, even by way of the pandemic.
“That is main for me and my household,” Grey stated two years after her therapy. “Two years with out me being within the hospital? Wow. We simply cannot imagine it. However we’re simply so grateful.”
Grey even began working full time promoting cosmetics at a Walmart, and commenced touring along with her husband, Earl.
A victory tour in London
NPR spent a day with Grey sightseeing in London earlier this yr throughout her first abroad journey. She had been invited to be the keynote speaker at a genetics convention celebrating the success of the therapy and exploring the remaining hurdles of gene-editing.
“The life that I as soon as felt like I used to be solely present in, I’m now thriving in,” Grey, who’s deeply spiritual, informed the viewers of scientists, medical doctors, ethicists and others, bringing many to tears. “I stand right here earlier than you in the present day as proof that miracles nonetheless occur and that God and science can coexist.”
Grey, who’s now 38, is particularly thrilled that she now not has to fret about dying prematurely from a stroke or different issues of sickle cell, leaving her kids with out a mom.
“The remedy has actually reworked my life greater than I might have ever imagined,” Grey stated. “It gave me a brand new lease on life.”
However she too frightened that many sickle cell sufferers will not be capable to get the therapy, together with a few of her personal relations who’ve the dysfunction. The therapy, which was developed by Vertex Prescription drugs and CRISPR Therapeutics, each in Boston, shall be marketed as Casgevy.
“That is a horrific feeling for me to have opened the door for sickle cell sufferers after which sickle cell sufferers do not obtain it,” Grey stated. “One thing must be accomplished to make sure that the sufferers who want it probably the most truly get it.”
